 It is used in the breading of food prior to frying to give the fried item a crisper flavour. In laboratories, NaHCO3 is routinely used to neutralise acid spills due to its mildly alkaline nature.In the kitchen, sodium bicarbonate serves several functions. To relieve constipation, a laxative made of sodium bicarbonate and polyethylene glycol dissolved in water is used before gastric and intestinal surgeries. It may be used to treat stomach or duodenal ulcer symptoms. It acts as a basic medium in the stomach, neutralising excess hydrochloric acid. The acid causes the bicarbonate to thermally decompose, releasing CO2 and causing the dough to rise and become fluffy and soft.Tartaric acid also serves another purpose in baking powder by preventing a metallic taste from the sodium carbonate formed during the chemical reaction.Īntacid: Another important application for sodium bicarbonate is as an antacid. It is mixed with cream of tartar (tartaric acid) to make baking powder, which is used in doughs to make cakes, bread, and other baked goods. Uses of NaHCO3:īaking soda: Sodium bicarbonate’s primary application is as baking soda. These conversions are important because NaHCO3 is used as a fire suppressant (“BC powder”) in some dry-powder fire extinguishers. Further heating (above 850 ☌/1,560 ☏) converts the carbonate to the oxide: This dehydration reaction occurs in the majority of bicarbonates. At 200 ☌ (392 ☏), the conversion is faster: Sodium bicarbonate gradually decomposes at temperatures ranging from 80–100 ☌ (176–212 ☏) into sodium carbonate, water, and carbon dioxide. NaHCO 3 + NaOH → Na 2 CO 3 + H 2 O Thermal decomposition: NaHCO 3 + CH 3 COOH → CH 3 COONa + H 2 O + CO 2 (g)Ĭarbonates are formed when sodium bicarbonate reacts with bases such as sodium hydroxide: When sodium bicarbonate reacts with acetic acid (found in vinegar), sodium acetate, water, and carbon dioxide are produced: One hydrogen ion and one bicarbonate ion combine to form a molecule of liquid water and a molecule of carbon dioxide gas, as seen above. The sign (aq), which stands for aqueous, indicates that the substance is dissolved in water (g) stands for gas, and (l) stands for liquid. As demonstrated below, bicarbonate ions react with acids. When sodium bicarbonate is dissolved in water, it separates into sodium (Na+) and bicarbonate ions, or charged particles (HCO3). These compounds’ modern chemical formulas now express their precise chemical compositions, which were unknown when the term “bicarbonate of potash” was coined. The prefix bi in bicarbonate derives from a pre-molecular naming system in reference to the two molar equivalents of carbon dioxide (known as carbonic acid in ancient chemistry) that potassium hydrocarbonate/bicarbonate releases upon decomposition to (di)potassium carbonate and potassium oxide (potash). 
Nomenclature:īecause it has been around for a long time and is widely used, the salt is known by many different names, including baking soda, bread soda, cooking soda, and bicarbonate of soda, and can be found in stores near baking powder. It is found as a precipitate in saline lakes and hot springs, among other places. Sodium bicarbonate occurs naturally in the form of nahcolite. Because of its alkaline nature, sodium bicarbonate has a slightly bitter taste. Other physical properties include the fact that it is odourless and water soluble. It is a crystalline compound that can also be found as fine powder. The fourth covalent bond is formed by carbon with an oxygen anion that is ionically bonded to a sodium cation.Īt room temperature, sodium hydrogen carbonate is a white solid. The bicarbonate ion is made up of a central carbon atom that is doubly bonded to oxygen and singly bonded to ahydrogen. Sodium bicarbonate molecules are made up of sodium cations and bicarbonate anions. 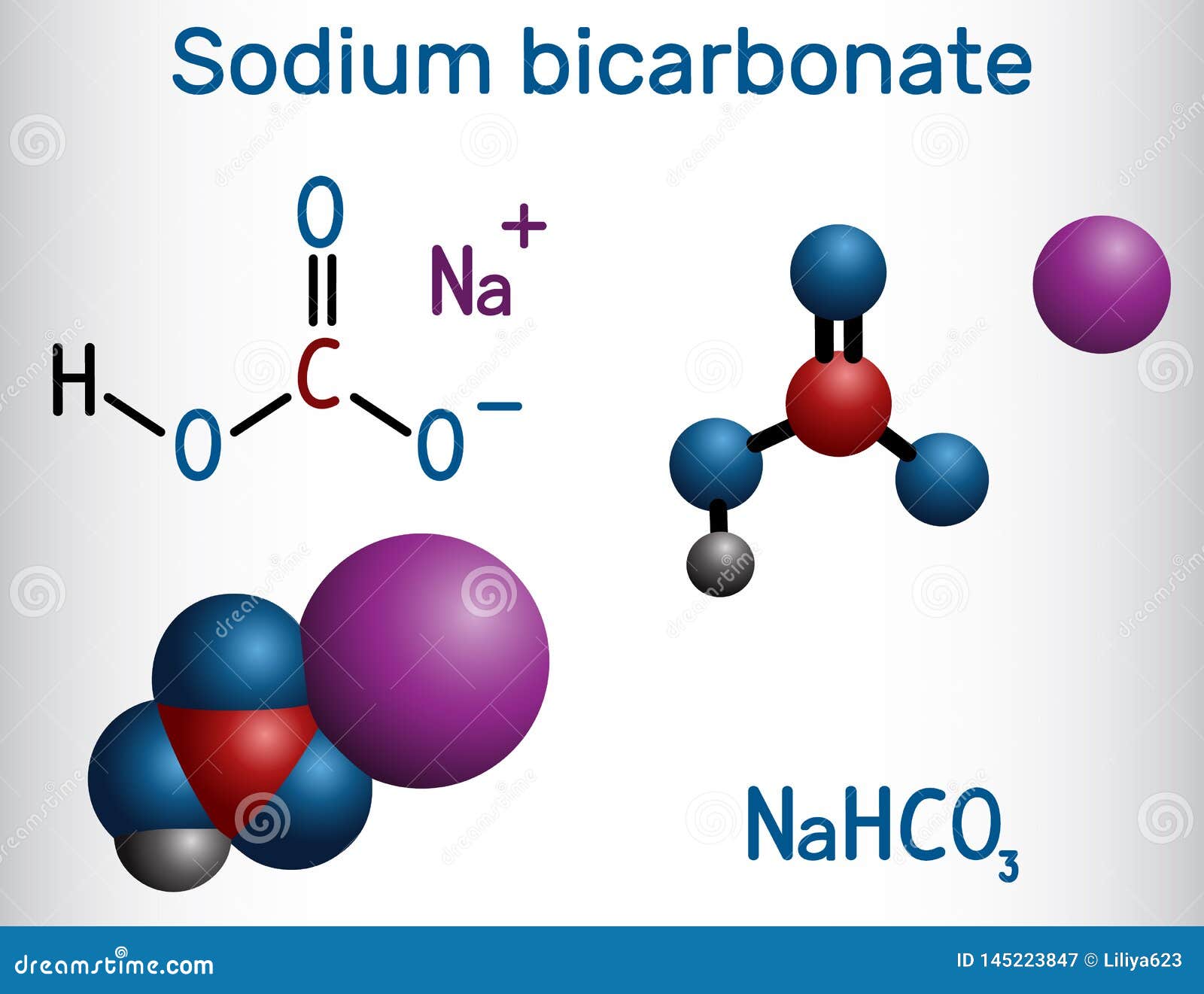
It is a constituent of the mineral natron and can be found dissolved in a variety of mineral springs. It has a slightly salty, alkaline flavour similar to washing soda (sodium carbonate). Sodium bicarbonate is a white crystalline solid that often appears as a fine powder. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
